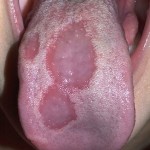
Lichen planus (LP) is an inflammatory condition that affects the skin and the squamous epithelium of mucosal surfaces lining the mouth, ears, eyes, and nose as well as the gastrointestinal and anogenital tracts. Two main patterns of inflammation are described: the plaque (raised) type and the erosive (raw) type, although bullous, blistering, or hypertrophic (thickened)types also occasionally occur.
The plaque type occurs most commonly and is estimated to affect up to 2% of the population. Erosive lichen planus (ELP) is a chronic, painful condition, which is often difficult to treat. The psychological, emotional, and physical distress associated with ELP affecting any mucosal site can be significant with affected individuals suffering low moods with or without treatment.
The main aim of this Cochrane review was to assess the effects of interventions in the treatment of erosive lichen planus affecting the oral, anogenital, and oesophageal regions.
A systematic search was conducted of the Cochrane Skin Group Specialised Register, the Cochrane Central Register of Controlled Trials (CENTRAL), Medline, Embase, and Lilacs databases. The reference lists of articles were also searched as well as online trials registries for ongoing trials. There were no language restrictions.
Randomised controlled trials (RCTs) that evaluated the effectiveness of any topical or systemic interventions for ELP affecting either the mouth, genital region, or both areas, in participants of any age, gender, or race were considered.
All 15 RCTs (473 participants in all) identified involved oral ELP only. Six of the 15 studies included participants with non-erosive lichen planus. In these studies, only the erosive subgroup was included for intended subgroup analysis. However, because of small numbers and the heterogeneity of the interventions, design methods, and outcome variables between studies it was not possible to pool data from any of the nine studies with only ELP participants or any of the six studies with the ELP subgroup.
One small study involving 50 participants found that 0.025% clobetasol propionate administered as liquid microspheres significantly reduced pain compared to ointment (Mean difference (MD) -18.30, 95% confidence interval (CI) -28.57 to -8.03), but outcome data was only available in 45 participants.
However, in another study, a significant difference in pain was seen in the small subgroup of 11 ELP participants, favouring ciclosporin solution over 0.1% triamcinolone acetonide in orabase (MD -1.40, 95% CI -1.86 to -0.94).
Aloe vera gel was 6 times more likely to result in at least a 50%improvement in pain symptoms compared to placebo in a study involving 45 ELP participants (Risk ratio (RR) 6.16, 95% CI 2.35 to 16.13).
In a study involving 20 ELP participants, 1% pimecrolimus cream was 7 times more likely to result in a strong improvement as rated by the Physician Global Assessment when compared to vehicle cream (RR 7.00, 95% CI 1.04 to 46.95).
There is no overwhelming evidence for the efficacy of a single treatment, including topical steroids, which are the widely accepted first line therapy for ELP. Several side-effects were reported, but none were serious. With topical corticosteroids, the main side-effects were oral candidiasis and dyspepsia.
They concluded
This review suggests that there is only weak evidence for the effectiveness of any of the treatments for oral ELP, whilst no evidence was found for genital ELP.
More RCTs on a larger scale are needed in the oral and genital ELP populations. We suggest that future studies should have standardised outcome variables that are clinically important to affected individuals. We recommend the measurement of a clinical severity score and a participant-rated symptom score using agreed and validated severity scoring tools. We also recommend the development of a validated combined severity scoring tool for both oral and genital populations.
Cheng S, Kirtschig G, Cooper S, Thornhill M, Leonardi-Bee J, Murphy R. Interventions for erosive lichen planus affecting mucosal sites. Cochrane Database of Systematic Reviews 2012, Issue 2. Art. No.: CD008092. DOI: 10.1002/14651858.CD008092.pub2.

[…] Dental Elf – 20th Feb 2012 – Only weak evidence of the effectiveness of any treatments f… […]