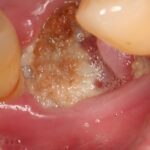
Necrosis of the jaws with exposed bone associated with bisphosphonate use was first reported in 2003 and is known as bisphosphonate-related osteonecrosis of the jaw (BRONJ). It has also been seen in patients taking antiresorptive and antiangiogenic medication and is termed medication-related osteonecrosis of the jaw. There is currently no consensus on treatment approaches which include non-surgical treatment non-surgical treatment, aggressive and conservative surgical treatments, drug holidays of bisphosphonates hyperbaric oxygen (HBO) antibiotics (ATB), teriparatides, autologous platelet concentrate (APC) therapy, and low-level laser therapy(LLLT).
The aim of this overview was to summarise the available evidence on the management and outcomes of medication-related osteonecrosis of the jaw (MRONJ)
Methods
Searches were conducted in the PubMed/Medline, Cochrane Library, Embase, Web of Science, LILACS, PROSPERO and OpenGrey databases. Systematic reviews of observational studies or meta-analysis of randomised clinical trials (RCTs) of surgical and non-surgical treatments for MRONJ were considered. Two reviewers independently searched for and screened the studies. Data abstraction was carried out independently by two reviewers and review quality assessed using AMSTAR 2 .
Results
- 15 reviews published between 2014 and 2020 were included
- The reviews covered 3 broad areas, conservative treatments, surgical treatments and alternative therapies.
- AMSTAR scores for the reviews ranged from 2 to 15
- 14 reviews evaluated whether surgical debridement was associated with antibiotics (ABT) or other adjuvant therapies with most concluded that conservative surgical treatment is effective only in the early stages of MRONJ.
- 7 reviews demonstrated that surgical resective treatment is more effective for the advanced stages of MRONJ
- 3 reviews reported insufficient evidence to alternative therapies.
- 5 of the reviews conducted meta-analyses with weak or inconsistent evidence.
Conclusions
The authors concluded: –
…there is weak evidence to support treatment protocols for MRONJ. Because of the low quality of the SRs, highlighted by this overview, no insightful therapeutic recommendations can be issued for any intervention for MRONJ. A higher number of clinical studies must be conducted in the future to support MRONJ therapies.
Comments
A broad database search was undertaken identifying 15 systematic reviews. In terms of evidence for management and treatment of MRONJ we should be looking for randomised controlled trials (RCTs) for good evidence for treatment. Of the included studies one review indicated 17 RCTs were included while the other included reviews only identified between 1-3 RCTs so seems like an outlier. Two Cochrane reviews were included in this review, the most recent one (Dental Elf -18th Oct 2017) including 5 RCTs rather than the 2RCTs indicted in the tables of this review. While the individual reviews a likely to have had a variable focus to the questions and different study inclusion criteria they were addressing and the numbers of the included studies in the reviews ranged from 97 to 1. As MRONJ is a relatively newly identified condition it is not unexpected that the numbers of high quality RCTs is thein on the ground. However, it would have been interesting if this overview had provided some indication of the number of unique RCTs available to guide treatment. As the authors acknowledge much of the literature related to MRONJ is derived from case reports, case series, and observational studies. In order to provide good evidence for the management of MRONJ high quality well-reported RCTs of appropriate sample size are needed to provide an evidence-based for future treatment.
Links
Primary Paper
Moraschini V, Calasans-Maia MD, Louro RS, Arantes EBR, Calasans-Maia JA. Weak evidence for the management of medication-related osteonecrosis of the jaw: An overview of systematic reviews and meta-analyses [published online ahead of print, 2020 Jun 26]. J Oral Pathol Med. 2020;10.1111/jop.13083. doi:10.1111/jop.13083
Other references
Dental Elf -18th Oct 2017
Medication-related osteonecrosis of the jaw (MRONJ): treatment and prevention
Photo Credits
By Coronation Dental Specialty Group – Own work, CC BY-SA 4.0, Link
