
Schizophrenia is a complex disorder (or, more likely, group of disorders) that has reality distortion at its core. Efforts to establish the cause of schizophrenia have been ongoing for more than a century, and many models have come and gone in that time (not for nothing has schizophrenia been called ‘the graveyard of neuropathologists’ (Plum, 1972)).
Part of the problem has been that very often the models would deal adequately with one aspect of the disorder (the neurochemistry, say, or the cognitive impairment) but fail to explain another, or just ignore it. Without a good model of schizophrenia, developing ways to treat the condition or prevent it before it begins is much harder, and much less likely to be successful.
In an article published in The Lancet at the end of last year, Oliver Howes and Robin Murray have set out what they call ‘an integrated sociodevelopmental-cognitive model’. As can no doubt be told from that title, this is a model that tries to account for the very wide range of factors that have been associated with increased risk for psychosis, from the neurobiological to the socio-demographic.

Howes and Murray bring together the dopamine hypothesis and the neurodevelopmental hypothesis in their new model
At root, this model suggests that the key biological dysfunction in people with schizophrenia is that they make and release too much of the neurotransmitter dopamine. This is not new (dopamine models of schizophrenia have been around since the first antipsychotics were shown to block dopamine receptors) but Howes and Murray tie this together with another well-established model, that of schizophrenia being a neurodevelopmental disorder.
In the original neurodevelopmental model, proposed by Murray (Murray and Lewis, 1987) and others (Weinberger, 1987) nearly 30 years ago, altered dopamine was a result of the interaction between early damage to frontal brain regions (perhaps as the result of complications during birth) and normal development.
Now, Howes and Murray argue events early in life, such as birth complications but also now including the effect of genes on brain development and childhood adversity, sensitise the dopamine system. This sensitisation makes the system more vulnerable to stress, such as that caused by social adversity. At the same time, cognitive models suggest that such stress can bias an individual’s thinking toward paranoia. A vicious circle develops, where the symptoms result in greater social adversity and stress, leading to more symptoms and so on, the end result being the full threshold psychotic break.
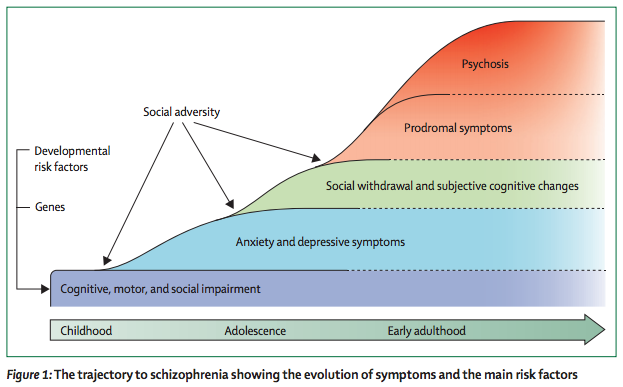
This model represents a real advance, in that it puts life events and the impact they have on the individual’s interpretation of the world around them at the centre of the process leading to schizophrenia. This fits with much of the advances of the past decade or so, that show that being an immigrant or being exposed to physical or sexual abuse are associated with a threefold increase in risk. It also suggests that interventions to reduce stress, or alter disordered patterns of thinking (such as cognitive behavioural therapy), ought to be particularly beneficial in the early phases of illness.
However, the article is not without problems. Largely, this is because the authors are not trying to conduct a systematic review, but to bolster the case for their model. In several places, the evidence is rather weaker than is made out, or is heavily reliant on animal models that may not in the end prove applicable to the human. Howes and Murray themselves concede that their model does not easily account for the persistent negative symptoms and cognitive impairments seen in many patients.
For me though, the major concern is whether a single model can ever fit a heterogenous disorder such as schizophrenia, or whether it is more likely to explain some fraction of the population with the diagnosis.
One example will serve to explain this point – Howes and Murray focus almost exclusively on dopamine as the neurotransmitter responsible for psychotic symptoms. However, we already know that many patients do not respond to treatment with traditional, dopamine-targeting, antipsychotics (Pilowsky, 1993). Indeed, there is evidence that non-responding patients show alterations in another neurotransmitter, glutamate, instead (Egerton, 2012). Still other reports indicate a subset of patients with changes to a specific group of acetylcholine receptors (Gibbons, 2013).
Better characterising the subgroups of patients with schizophrenia is a critical condition of establishing the success or failure of this new model.

There is no ‘one size fits all’ solution for people with schizophrenia
Links
Howes OD and Murray RM. Schizophrenia: an integrated sociodevelopmental-cognitive model. The Lancet – 6 December 2013. DOI: 10.1016/S0140-6736(13)62036-X. [Abstract]
Plum F. Prospects for research on schizophrenia. Neuropathological findings. Neurosci Res Prog Bull. 1972;10:384–388. [PubMed abstract]
Murray RM and Lewis. Is schizophrenia a neurodevelopmental disorder? British Journal of Psychiatry 1987;295:681-682.
Weinberger DR. Implications of Normal Brain Development for the Pathogenesis of Schizophrenia. Archives of General Psychiatry 1987;44:660-669. [PubMed abstract]
Pilowsky LS et al. Antipsychotic medication, D2 dopamine receptor blockade and clinical response: a 123I IBZM SPET (single photon emission tomography) study. Psychological Medicine 1993;23:791-797. [PubMed abstract]
Egerton et al. Anterior cingulate glutamate levels related to clinical status following treatment in first-episode schizophrenia. Neuropsychopharmacology 2012;37:2515-2521.
Gibbons et al. Widespread decreases in cortical muscarinic receptors in a subset of people with schizophrenia. 2013;16:37-46. [PubMed abstract]


RT @Mental_Elf: A new model for schizophrenia? http://t.co/0ZetiWuCwe
@Mental_Elf we have been saying 4 years! life events&resulting thinkings affecting physiological processes & resulting pathology from social
A new model for schizophrenia?: Schizophrenia is a complex disorder (or, more likely, group of disorders) that… http://t.co/oGSE4Ob7UJ
Charlotte Rowe liked this on Facebook.
Interesting points made, attempt to be all inclusive.
Tu as vu le colloque a Bordeaux vers le 20 juin? 5 journees dont une sur la schz et une sur les toc. Entre autres…
Paula Gardiner liked this on Facebook.
Pagan Warren liked this on Facebook.
Surely this is another sign that it’s time to dump schizophrenia as a label. It no longer fits with what we know.
A new model for schizophrenia? http://t.co/GneCgaucPa from the always excellent @Mental_Elf
Today @StephenWood8 discusses the new integrated sociodevelopmental-cognitive model for schizophrenia http://t.co/AmOaohqduT
Oooh, this is excellent! #PSYa4 Causes of Sz. A bit like a diathesis-stress model.
A new model for schizophrenia? http://t.co/Q15zJcNzVJ
A new model for schizophrenia? – The Mental Elf http://t.co/PVvHksD0cC
Mental Elf: A new model for schizophrenia? http://t.co/hjAp8JGvYU
The Mental Elf liked this on Facebook.
Bespoke Training Services UK Ltd liked this on Facebook.
Being an immigrant or being exposed to physical/sexual abuse are assoc w/ 3-fold increase in risk of schizophrenia http://t.co/AmOaohqduT
. @Mental_Elf that sounds like coincidental correlation to me!
Read our summary of @TheLancet review by Oliver Howes & Robin Murray, which presents a new model for schizophrenia http://t.co/AmOaohqduT
MT: Immigrants & physical/sexual abuse survivors have 3-fold increase in risk of schizophrenia http://t.co/O0I9pWPHLl http://t.co/B0i4KOLOtC
Thought provoking.. http://t.co/BOqc6tYdCi
Don’t miss: A new model for schizophrenia? http://t.co/AmOaohqduT
@Mental_Elf hardly new stress vulnerability +
Via @Mental_Elf A new model for schizophrenia? http://t.co/w2wCOOFTqd Robin Murray tries to include trauma without giving up dx & dopamine
@Mental_Elf Do you ever research the demonic influence on schizophrenia?
This where the demonic takes up host in the affected person.
RT @Mental_Elf: New schizophrenia model brings together the dopamine hypothesis & the neurodevelopmental hypothesis http://t.co/AmOaohqduT
A new model for schizophrenia? – The Mental Elf – http://t.co/cT2sjV8Vaq umm?
João Leal liked this on Facebook.
Anneka Holden liked this on Facebook.
Anyone with a knowledge of what symptoms, signs and syndromes are, and is prepared to examine schizophrenia critically will tell you it is not a brain disease.
Great @Mental_Elf summary by @StephenWood8 of important paper synthesising biological & social schizophrenia models http://t.co/SO7Pc3mpqO
A new model for schizophrenia? http://t.co/4ZZ4UxkdaN vía @Mental_Elf
Lydia Watson liked this on Facebook.
Interesting discussion in @Mental_Elf website about schizophrenia: http://t.co/vfwz5VYBYK
Im wondering about maternal stress, wether this affects the foetus in utero, the developing brain, and sensitivity to dopamine/glutamate?
[…] https://www.nationalelfservice.net/mental-health/schizophrenia/a-new-model-for-schizophrenia/ […]