
Hot on the tail (in guideline terms) of the NICE bipolar disorder guidelines [NICE 2014] (National Collaborating Centre for Mental Health, 2014) comes the evidence-based guidelines for treating bipolar disorder: Revised third edition recommendations from the British Association for Psychopharmacology [BAP 2016] (Goodwin et al., 2016).
Here I’ll examine the major differences between these two guidelines, and why these may have occurred. I’ll then look at pharmacological treatment recommendations for bipolar disorder (BPD) from the new BAP guidance. Due to the length of the guidance, only key points are raised in this blog.
BAP 2016 and NICE 2014 offer different interpretations of the evidence for treatment of bipolar disorder.
Methods
Respective authors updated subsections of the guidance from the 2009 version (Goodwin et al., 2009). The NICE 2014 guideline was considered a comprehensive collation of all relevant data up until 2012-2013, and was augmented by MEDLINE searches to December 2015.
Results
Major differences: NICE 2014 vs BAP 2016
Assessment of the evidence base
Unlike NICE 2014, previous editions (Goodwin & Young., 2003; Goodwin et al., 2009) and other BAP guidelines, BAP 2016 did not follow the traditional I to IV hierarchical evidence categories (i.e. I = randomised controlled trials [RCTs], IV = opinion/clinical impression). The authors state this is because:
This approach explicitly downgrades non-experimental descriptive studies of treatment effects in favour of any randomised controlled trial; in so doing, it confounds design with quality.
They instead employed an approach based on the Cochrane Collaborations GRADE system. This is an important departure from seeing all RCTs as ‘gold standard’, when key issues of generalisability often limit their interpretation. They therefore downgraded findings from small, inconclusive RCTs and upgraded findings from observational studies in large samples with strong quasi-experimental designs. This approach is likely to become increasingly important as large amounts of digital patient data become available to researchers. More can be read about this in a recent New England Journal of Medicine article (Bothwell et al., 2016).
Whereas NICE 2104 downgrades RCTs with high drop-out, BAP 2016 considers it common and a “limitation of RCTs in general”. BAP 2016 also considered evidence from longer-term relapse prevention studies, discounted by NICE, especially if results of naturalistic studies corroborated RCT findings. Therefore, in general, BAP 2016 was more inclusive of available evidence.
One way in which they were more cautious was in the interpretation of the “league table” of treatments that can be derived from network meta-analyses (see Cipriani et al., 2011 for a relevant example). Goodwin et al. argue that this is because confidence intervals around rankings are wide and that “it does not follow that practice be dominated by evidence that one effective treatment is, on average, slightly better than another”.
Drug names
Another deviation from previous editions (and from NICE 2014) is the use of Neuroscience-based-Nomenclature (NbN). The aim of this system is to avoid generic terms for drugs based on indication and instead use descriptions of mode of action, which should help clinicians to make more informed treatment options. However, there are problems with this approach, especially in the treatment of bipolar disorder. For example, not all “dopamine receptor antagonists” can be considered equal and it can make reading the guidance more difficult.
There is likely to be ongoing discussion about the use of NbN amongst academics, clinicians and patients. This recent Lancet Psychiatry Editorial provides a useful summary (The Lancet Psychiatry, 2016).
Diagnostic approach
BAP 2016 recommends use of the Diagnostic and Statistical Manual version 5 (DSM-5) criteria for bipolar disorder, whilst NICE 2014 refers to both DSM and International Classification of Disease 10th edition (ICD-10). NICE goes into some detail about the differences between these classification systems, with regards to bipolar disorder. In summary, ICD-10 criteria for diagnosis of bipolar disorder are stricter, as are criteria for defining mania vs hypomania. There is also no I/II differentiation in ICD-10.
These differences will lead to more individuals being diagnosed under DSM criteria than ICD, and this may partially explain worldwide differences in prevalence and incidence estimates of bipolar disorder.
Access to care
NICE 2014 suggests that the treatment needs of individuals with bipolar disorder can be met by existing service models, including those that are stable being returned to the care of their GP. BAP 2016 disagrees with this and identifies a specific need to extend early intervention services, provided by experts in bipolar disorder.
Fragmentation of NHS services between ‘inpatient’/’outpatient’, ‘assessment’/’treatment’ and ‘psychotic’/’non-psychotic’ may have had a differentially adverse impact on individuals with bipolar disorder, who often (in my clinical experience) fall between services.
Adolescent diagnosis
BAP 2016 is eager to guard against inappropriate diagnosis in children and adolescents, however the authors are also keen to avoid delayed diagnosis in those clearly fulfilling criteria. They believe the wording in NICE 2014 (“diagnosis… should be made only after a period of intensive, prospective longitudinal monitoring”) could lead to inappropriate delays in receiving treatment. As with the diagnosis of schizophrenia, it is likely that many clinicians are reluctant to make an early diagnosis, however unlike schizophrenia, these individuals won’t have access to a ‘diagnosis’ (i.e psychosis) that leads to engagement with appropriate services and treatments. The diagnostic delay in bipolar disorder is well-recognised (Saunders & Goodwin, 2010). However, there will always be a tension between treating those who did not need care and not treating those who did.
Psychological interventions
BAP 2016 focus (perhaps unsurprisingly) on pharmacological treatments for bipolar disorder. The lead author (Guy Goodwin) wrote a blog post for The Mental Elf earlier this year reviewing an article (Jauhar et al. 2016) describing the potential overselling of psychosocial interventions, perhaps resulting from author bias. This issue is further addressed in BAP 2016, with the authors concluding that they: “have favoured caution in interpreting the findings from psychosocial interventions” and that “[r]ecommendations for psychotherapy alone (as in NICE 2014…) are surprising”.
Authorship
It should be noted that of the BAP 2016 authors, only two were on the NICE 2014 development group. Confusingly, one of the authors of both guidelines has argued for the mandatory implementation of NICE 2014 guidance in the National Health Service (Morriss, 2015).
Patient choice/prescribers choice
In perhaps their most confrontational mode, the authors state:
[D]rug choice is an important clinical freedom… While NICE 2014 is at pains to emphasize the need to respect patient choice, as are we, they are less liberal in what choices they actually sanction.
Unless, of course, your choice is for a psychological intervention!
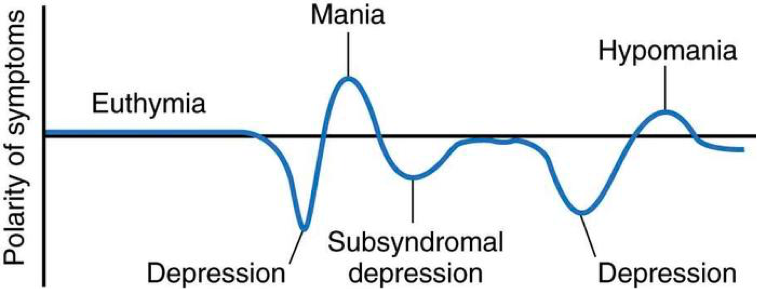
Treatment of bipolar disorder is complex because of the different phases of illness
Treatment recommendations
Treatment of acute manic episodes
Both guidelines agree on the appropriate treatment of mania:
- Haloperidol, olanzapine, risperidone and quetiapine are identified as having the highest efficacy
- Combinations of antipsychotics with lithium or valproate can be superior to lithium or valproate alone and should be considered especially when breakthrough mania with one agent has occurred
- Discontinuation of treatments can be considered after full remission (often of the order of 12 weeks)
- Treatment choice should be dictated by the clinical context and patient preference
Short-term treatment of depression
NICE 2014 and BAP 2016 disagree on depression treatment. Goodwin et al state:
- Quetiapine has the most convincing efficacy for short-term and relapse prevention
- Olanzapine (combined with fluoxetine, or as monotherapy), Lurasidone (not licenced for use in bipolar depression in Europe) and lamotrigine have evidence of efficacy
- Antidepressants may be effective [italics authors own] but require co-prescription of a mood stabilising agent
- Risk of a switch to mania is higher in tricyclic antidepressants and other duel-action medications (e.g. venlafaxine) than in SSRI
- Lithium, valproate and carbamazepine have poor evidence for acute efficacy
- Consider ECT in severe or refractory depression
- Consider family focused, cognitive behavioural therapy or interpersonal rhythm therapy as an additional treatment
- Consider a strategy for long-term treatment as the patient recovers
The main disagreements are that NICE suggest fluoxetine has a ‘special’ nature amongst antidepressants, that use of psychological treatments alone may be appropriate and that ranking of drugs from network meta-analysis can be relied upon.
Long-term mood stabilisation
There is broad agreement between NICE 2014 and BAP 2016 when it comes to longer-term management, with BAP 2016 offering more treatment options:
- Lithium remains the most effective treatment – preventing relapse and reducing suicide risk, and should be considered for all patients willing to take it reliably
- Lamotrigine is effective against depression in long-term treatment
- Antipsychotic medications reduced the risk of relapse and response may be predicted by acute treatment effects
- Valproate has limited trial data, and is less effective than lithium
- Carpemazepine is less effective than lithium
- If there has been an acute response to antidepressants these can be continued if risk of depressive relapse is high. They should be used in conjunction with an anti-manic medication
- Combination drug treatments are likely to be necessary, but RCTs of monotherapy vs combinations are rare
- Psychoeducation is the ‘first-line’ psychological intervention, with superior benefits early in the course of illness

Recent research has strengthened the position of lithium as the first-line mood stabiliser.
Strengths and limitations
There is much to be praised in BAP 2016:
- It offers a broader range of drug treatment options than NICE 2014, with detailed discussion of the evidence base
- It achieves a more accurate analysis of the strengths and limitations of the evidence base for each medication than NICE 2014
- It appreciates the complicated nature of applying “evidence based treatment” to a heterogeneous and highly complex group
- It provides a good overview of the appropriate treatment of special groups and situations (i.e. bipolar disorder plus alcohol and substance misuse disorder, bipolar disorder and borderline personality disorder, bipolar disorder and anxiety, bipolar disorder and pregnancy), which has not been covered in this blog
- It offers a succinct guide to commencing and monitoring commonly used drugs
- Like NICE 2014, it provides recommendations for future research which will guide research priorities
Limitations are:
- In the availability of evidence that can inform guidance, especially in the treatment of bipolar depression and the use of psychotherapy
- BAP 2016 may have gone too far (perhaps in an attempt to provide a counterpoint to NICE 2014) in dismissing psychological therapy
- The use of NbN can make reading the guidelines less, rather than more, straight forward
- The use of DSM criteria may complicate application to countries that use ICD (such as the UK)

Psychoeducation is recommended by both BAP 2016 and NICE 2014.
Summary
BAP 2016 provides a wider range of treatment options to clinicians and patients, this is very important given the calls for mandatory implementation of guidance from NICE 2014, which is relatively restrictive. It appears to be a more appropriate interpretation of the available evidence for medications and as such is particularly important for clinicians prescribing drug treatments.
The BAP reading of the evidence for psychotherapies is more conservative than NICE, though both advocate psychoeducation. There is a lack of quality evidence about the (positive and potentially negative) effects of psychological interventions in bipolar disorder.

There’s a lack of quality evidence about the effects of psychotherapies for bipolar disorder.
Conflicts of interest
Joseph Hayes has published studies on the effectiveness and adverse effects of longer-term drug treatments for bipolar disorder. He has worked with a number of the authors of this BAP Guideline in the past. He has never received drug company funding. He last ate a sandwich provided by a drug company eight years ago.
Links
Primary paper
Goodwin GM, Haddad PM, Ferrier IN. et al (2016) Evidence-based guidelines for treating bipolar disorder: Revised third edition recommendations from the British Association for Psychopharmacology (PDF). Journal of Psychopharmacology. 2016 30(6):495-553.
Other references
American Psychiatric Association. Diagnostic and statistical manual of mental disorders (DSM-5®). American Psychiatric Pub; 2013 May 22.ICD-10
Bothwell LE, Greene JA, Podolsky SH, Jones DS. (2016) Assessing the Gold Standard—Lessons from the History of RCTs. New England Journal of Medicine. 2016 374(22):2175-81.
Cipriani A, Barbui C, Salanti G, et al (2011) Comparative efficacy and acceptability of antimanic drugs in acute mania: a multiple-treatments meta-analysis. The Lancet. 2011 378(9799):1306-15.
Goodwin GM &Young AH (2003) The British Association for Psychopharmacology guidelines for treatment of bipolar disorder: a summary. Journal of Psychopharmacology. 2003 (4 Suppl):3-6.
Goodwin GM & Consensus Group of the British Association for Psychopharmacology (2009) Evidence-based guidelines for treating bipolar disorder: revised second edition—recommendations from the British Association for Psychopharmacology. Journal of Psychopharmacology. 2009 23(4):346-88.
Jauhar S, McKenna PJ, Laws KR. (2016) NICE guidance on psychological treatments for bipolar disorder: searching for the evidence. The Lancet Psychiatry. 2016 Apr 30;3(4):386-8. [PubMed abstract]
The Lancet Psychiatry (2016) Naming names. The Lancet Psychiatry. 2016 3(6):489. [PubMed abstract]
Morriss R. (2015) Mandatory implementation of NICE Guidelines for the care of bipolar disorder and other conditions in England and Wales. BMC medicine. 2015 Sep 30;13(1):1.
National Collaborating Centre for Mental Health (2014). Bipolar Disorder: The Management of Bipolar Disorder in Adults, Children and Adolescents. Leicester, England: British Psychological Society; 2014. NICE Clinical Guidelines CG185
Saunders KE, Goodwin GM (2010) The course of bipolar disorder (PDF). Advances in Psychiatric Treatment. 2010 16(5):318-28.
World Health Organisation. The ICD-10 classification of mental and behavioural disorders: clinical descriptions and diagnostic guidelines, Geneva: World Health Organization; 1992.

@Mental_Elf really engaging blog @in_psych agree with lack of information about potential negative effects of psychological interventions
@Mental_Elf @LannonElizabeth ?
This is a good clear summary of the BAP guideline on the treatment of Bipolar Disorder. The BAP guideline itself provides a much needed balance to the bias inherent in the latest NICE guideline on treatment of bipolar disorder – principally with regard to psychological interventions – see https://theconversation.com/are-treatment-guidelines-on-schizophrenia-and-bipolar-disorder-just-plain-wrong-54369
As the BAP guideline states “…the prominent endorsement of psychological treatments for bipolar disorder, without qualification, as ‘Key priorities for implementation’, goes well beyond the evidence. The partial way in which the data appears to have been reviewed by NICE2014 to justify their conclusions has also been highlighted (Jauhar, McKenna and Laws 2016).” – see http://thelancet.com/journals/lanpsy/article/PIIS2215-0366(15)00545-3/abstract
The BAP are rightly sceptical about psychological interventions for bipolar disorder – they uniformly lack evidence. However, I would however raise one question regarding the BAP’s endorsement of psychoeducation. It seems to me that evidence for psychoeducation is similarly lacking (see Jauhar, McKenna and Laws 2016).
For example, in the latest NICE guideline:
Two meta-analyses (each with 2 trials) reported that online psychoeducation produced no significant symptom reduction compared with treatment as usual, either at post-treatment (SMD −0·18, −0·63 to 0·26) or follow-up (SMD −0·36, −1·09 to 0·37). Group psychoeducation likewise had negative results compared with treatment as usual at post-treatment (SMD 0·14, −0·17 to 0·46, two trials) and follow-up (SMD 0·40, −0·07 to 0·87, one trial).
Family psychoeducation was reported to be beneficial at post-treatment compared with treatment as usual (SMD −0·73, −1·35 to −0·10) and with active control (SMD −0·40, −0·80 to −0·00), but family psychoeducation was not reported as beneficial at 12 months follow-up compared with treatment as usual (SMD −0·15, −0·69 to 0·39), or with active control (SMD −0·10, −0·56 to 0·36); however, all these three meta-analyses contained only one study each.
So I am somewhat curious about the BAP’s endorsement of psychoeducation
The group’s endorsement of psychoeducation appears most prominently in section 3 on enhanced care (p 23). In fact, many of the recommended elements of psychoeducation are in effect regarded as standard of care and so based in large measure on that much down-graded category of evidence, expert opinion. The expert consensus was that patients needed to know about their illness and what they could do about it, and that ‘psychoeducation’ could never be truly tested against ‘psychoignorance’. While it is not quite like testing the efficacy of a parachute, it was rather in that direction.
In discussing the underlying evidence driving the recommendation and in grading psychoeducation as ‘4 star’, we put particular emphasis on the open randomised trial in young patients conducted in Denmark (Kessing LV, Hansen HV, Hvenegaard A, et al. (2013) Treatment in a specialised out-patient mood disorder clinic v. standard out-patient treatment in the early course of bipolar disorder: Randomised clinical trial. Br J Psychiatry 202: 212–219), which the group found very compelling. I believe that we fairly discuss the limitations of much of the other literature; it is generally compatible with the interpretation that psychoeducation is likely to be helpful when offered early in the illness course and less so later. Of course, for convenience most of the trials have over-recruited more mature patients, who simply may not have a lot to learn.
Hi Keith,
Thank you for this very clear summary of the limited/lack of evidence for psychoeducation. BAP 2016 is pretty cutting about the guidance for psychological interventions in NICE 2014 and I wonder if, as you are saying, they should have gone further.
I wonder if psychoeducation trials fail to show between group differences because treatment as usual includes a good dose of psychoeducation anyway? That is, trials are not psychoeducation vs. psychoignorance. However, this obviously means we can’t justify funding specialist services to provide psychoeducation with the evidence base as it stands.
Recent research has strengthened position of #lithium as first-line mood stabiliser for #bipolar disorder https://t.co/qANIqKZrJk
RT @Mental_Elf: Treatment of bipolar disorder is complex because of the different phases of illness https://t.co/qANIqKZrJk https://t.co/OL…
RT @Mental_Elf: There’s a lack of high quality evidence about psychotherapies for bipolar disorder @in_psych on new BAP guidance https://t.…
RT @Mental_Elf: Today @in_psych on the British Association for Psychopharmacology guidance on treatment for bipolar disorder https://t.co/q…
RT @in_psych: My first @Mental_Elf blog: BAP guidelines for treating bipolar disorder https://t.co/gY4xi5aN1L
RT @Mental_Elf: Psychoeducation for bipolar disorder is recommended by both BAP 2016 and NICE 2014 https://t.co/qANIqKZrJk
#Psychoeducation is recommended for bipolar disorder by @NICEcomms & @BAPsych Good discussion taking shape at https://t.co/qANIqKZrJk
Don’t miss: Evidence-based guidelines for treating #bipolar disorder https://t.co/qANIqKZrJk
Evidence-based guidelines for treating bipolar disorder https://t.co/RdXNQaAXgt @Mental_Elf https://t.co/kRCVWHt3Ls
Evidence-based guidelines for treating bipolar disorder https://t.co/hmv3ScNA1x via @sharethis
RT @stressdev: Clear and helpful @Mental_Elf blog on @BAPsych guidelines for #bipolardisorder treatment by @in_psych https://t.co/9xFIyDCh8n
@Mental_Elf TY for that! I am just about to write about my experiences of being antipsychotic free for the first time in 5 years and what…
@Mental_Elf …I feel quetiapine has and has not done for me over that period. So useful to see evidence on efficacy.
@Mental_Elf My consultant is wonderful. She’s implemented all stated here including EI & frequent, ongoing support. This was a great read??
@Mental_Elf …to great effect may I add. ?
RT @Mental_Elf: Don’t miss: Evidence-based guidelines for treating #bipolar disorder https://t.co/G2N8TUlALy
Most popular blog this week?
It’s @in_psych
Evidence-based guidelines for treating #bipolar disorder
https://t.co/qANIqKZrJk
@Mental_Elf @stokeymummy @in_psych useful read @PCMHbps
Evidence-based guidelines for treating bipolar disorder https://t.co/FUTdf7P6Nv